There are 230 types of space groups. These are not unrelated to each other; they can be classified (grouped) by focusing on shared properties. Concepts such as crystal systems and Bravais lattices are also methods of classifying space groups, but our previous explanations were somewhat imprecise or incomplete. Here we provide a detailed explanation of several commonly used classification schemes for space groups, proceeding from larger classifications (more categories) to smaller classifications (fewer categories).
Space Groups and Space Group Types
Throughout this website, we have repeatedly written statements like “there are 230 types of space groups,” but this is actually an imprecise expression. More precisely, there are 230 space group types. A space group necessarily includes translation operations, and the translation amounts and directions differ from crystal to crystal. For example, both NaCl and MgO have the symmetry of the space group \(Fm\bar{3}m\), but their lattice parameters differ (the former has a=5.64Å, the latter a=4.21Å), so even though the notation is the same, they must be considered as belonging to space groups with different symmetry operations. In other words, an infinite number of space groups exist.
On the other hand, if we organize space groups by their algebraic structure while ignoring differences in lattice parameters, we obtain 230 space group types. For most readers, “ignoring differences in lattice parameters” when discussing space groups is probably an almost self-evident assumption. Therefore, on this website, with a slight sacrifice of precision but within a range where no misunderstanding arises, we will simply refer to space group types as space groups1.
Affine Space Groups
As described on the “2.4. Group Multiplication Tables and Isomorphism” page, among the 230 three-dimensional space groups, the following 11 pairs are isomorphic to each other.
- Tetragonal: \(P4_1\) and \(P4_3\), \(P4_122\) and \(P4_322\), \(P4_12_12\) and \(P4_32_12\)
- Trigonal: \(P3_1\) and \(P3_2\), \(P3_121\) and \(P3_221\), \(P3_112\) and \(P3_212\)
- Hexagonal: \(P6_1\) and \(P6_5\), \(P6_2\) and \(P6_4\), \(P6_122\) and \(P6_522\), \(P6_222\) and \(P6_422\)
- Cubic: \(P4_132\) and \(P4_332\)
Each pair has an enantiomorphic relationship. The 219 space groups obtained by treating these 11 isomorphic pairs as identical are called affine space groups (affine space-group types). On the other hand, the 230 types that count isomorphic duplicates as independent are called proper affine space groups (proper affine space-group types) or crystallographic space groups (crystallographic space-group types). Usually, when we say “space groups,” we refer to the latter.
Crystal Classes and Crystallographic Point Groups
As described on the “2.4. Subgroups and Cosets” page, when we derive the quotient group modulo the translation group for every space group, each corresponds to one of the 32 crystallographic point groups (point groups formed by combining rotations and rotoinversions compatible with translations). This classification that assigns space groups to point groups is called a crystal class2 or a geometric crystal class. Crystal classes and crystallographic point groups have a one-to-one correspondence and their notations are exactly the same, but please note that the concepts are subtly different. The former refers to the point group corresponding to a space group, while the latter refers to the point group itself.
In general, crystal classes are deeply related to the macroscopic properties of crystals. This is because crystal classes correspond to the classification of macroscopic crystal morphology according to morphological symmetry. For example, the external shape of a crystal and various physical properties (elastic modulus, thermal conductivity, magnetic permeability, etc.) follow the point group symmetry expressed by the crystal class.
Which crystal class a given space group belongs to can actually be determined by the following simple symbol substitutions and deletions. Namely, from the space group notation (Hermann-Mauguin notation):
- Delete the first character (the lattice symbol)
- Delete all subscript numbers (translational information of screw axes)
- Convert all lowercase alphabetic characters (glide operations) to \(m\)
For example, the crystal class of the space group \(Fd\bar{3}m\) is \(m\bar{3}m\), and the crystal class of the space group \(P6_422\) is \(622\).
Arithmetic Crystal Classes and Symmorphic Space Groups
When we simply say “crystal class,” we refer to the geometric crystal class described above. There is actually another similar term: arithmetic crystal class. An arithmetic crystal class is a concept that combines the (geometric) crystal class with the Bravais lattice. While there are 32 crystal classes, there are 73 arithmetic crystal classes. Arithmetic crystal classes have a one-to-one correspondence with symmorphic space groups. A symmorphic space group is one that contains no screw or glide symmetry operations, and is formed solely by combining point group operations with lattice translations. For example, the space group \(Fm\bar{3}m\) is a symmorphic space group, but \(I4_1acd\) is not.
Bravais Lattices and Bravais Arithmetic Classes
As already explained on another page, the Bravais lattice (Bravais lattice, Bravais type of lattice3) is a classification based on the idea: if we assume a crystal where atoms exist only at the vertices of a primitive lattice and vary the lattice shape in various ways, how many types of space groups can represent it? Below, we reproduce the 14 types of Bravais lattices and additionally list the corresponding space groups (the overall symmetry of the arranged lattice) and point groups (the symmetry that keeps one lattice point fixed).
| Symbol | Schematic | Space group | Point group |
|---|---|---|---|
| \(aP\) | 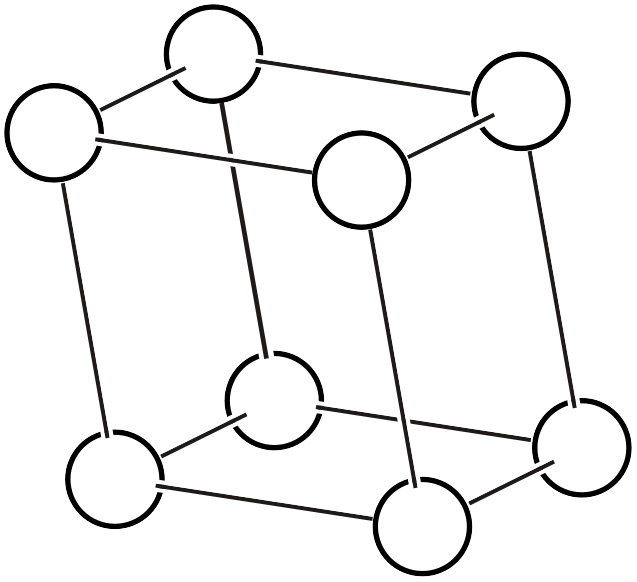 | \(P\bar{1}\) | \(\bar{1}\) |
| \(mP\) | 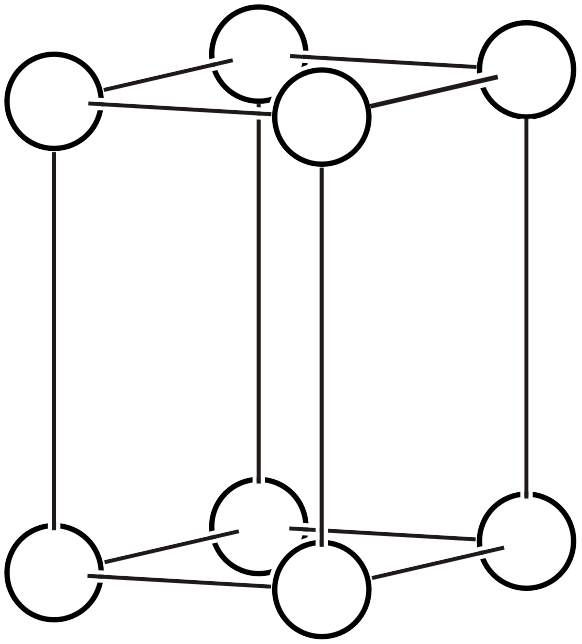 | \(P2/m\) | \(2/m\) |
| \(mC\) | 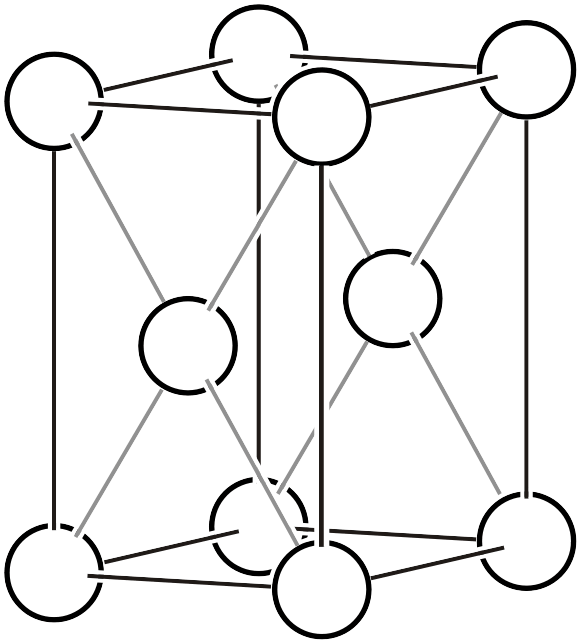 | \(C2/m\) |
| Symbol | Schematic | Space group | Point group |
|---|---|---|---|
| \(oP\) | 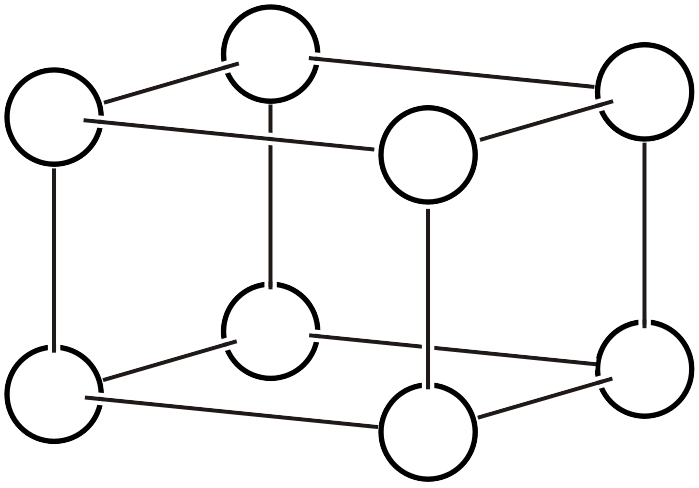 | \(Pmmm\) | \(mmm\) |
| \(oC\) | 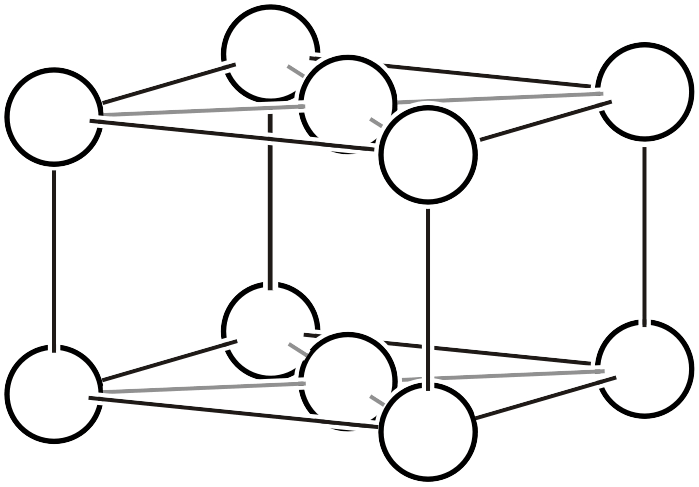 | \(Cmmm\) | |
| \(oI\) | 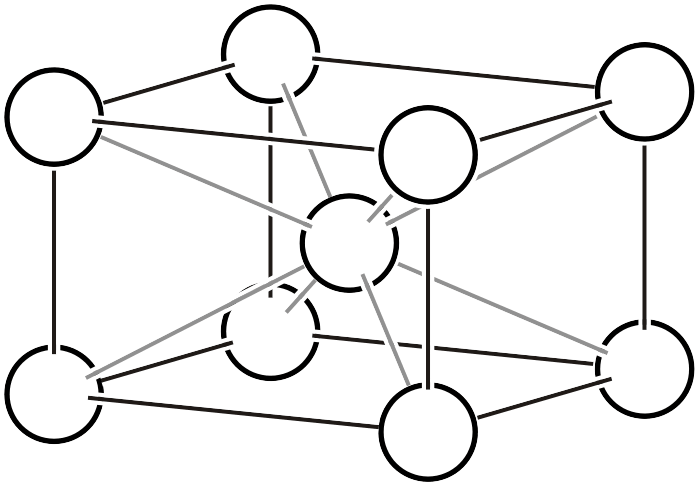 | \(Immm\) | |
| \(oF\) | 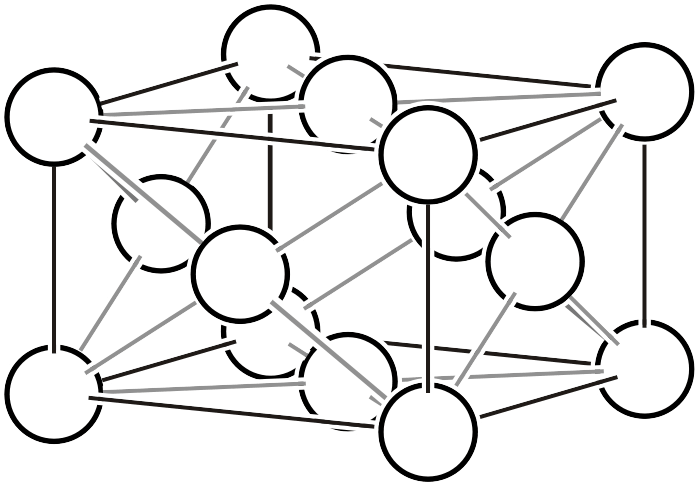 | \(Fmmm\) | |
| \(tP\) | 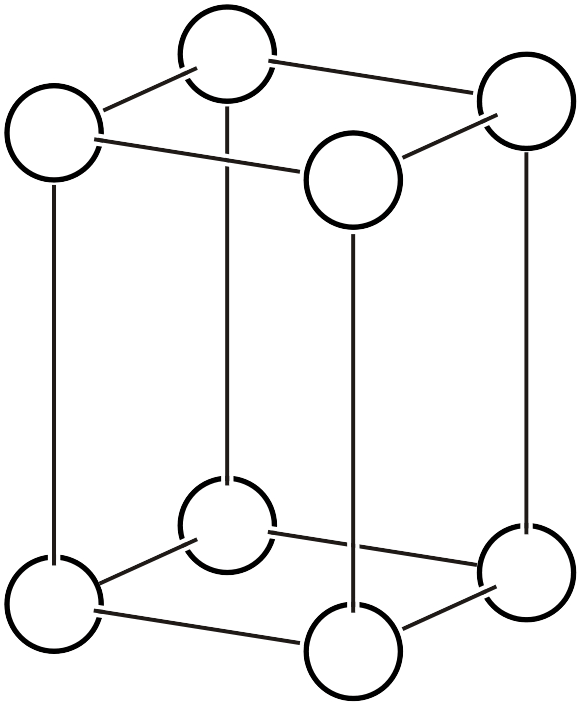 | \(P4/mmm\) | \(4/mmm\) |
| \(tI\) | 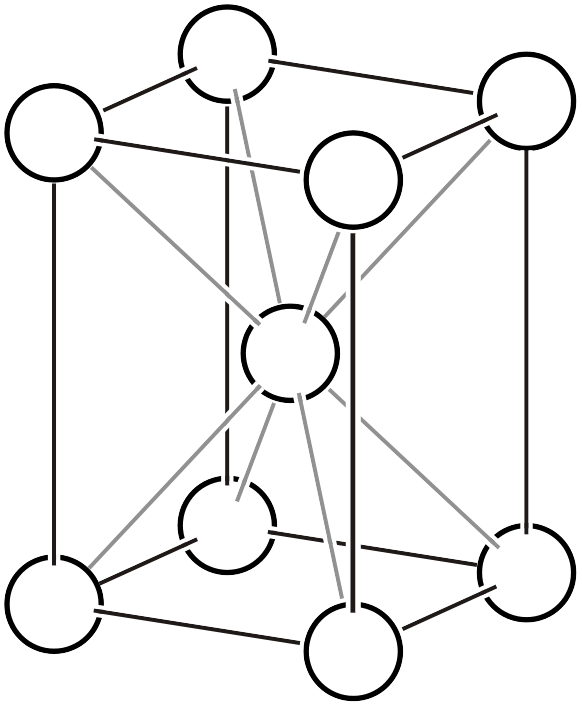 | \(I4/mmm\) |
| Symbol | Schematic | Space group | Point group |
|---|---|---|---|
| \(hR\) | 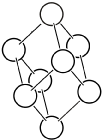 | \(R\bar{3}m\) | \(\bar{3}m\) |
| \(hP\) | 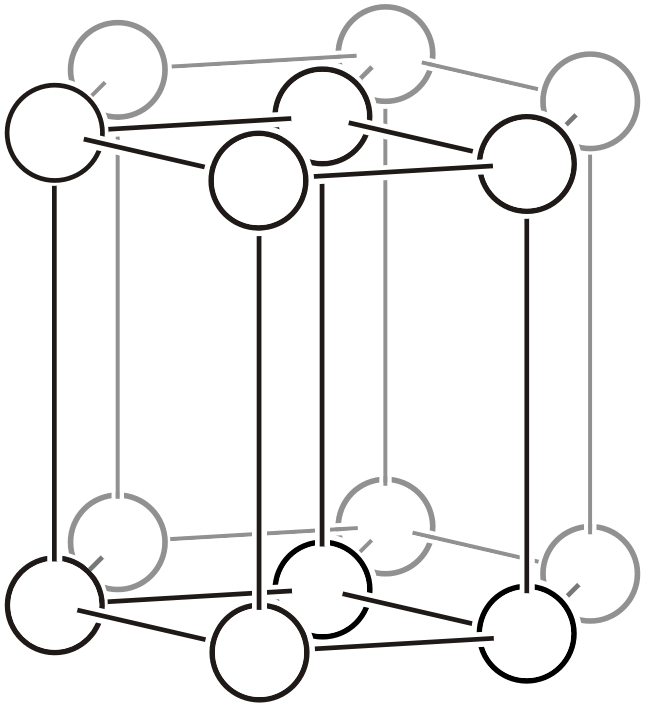 | \(P6/mmm\) | \(6/mmm\) |
| \(cP\) | 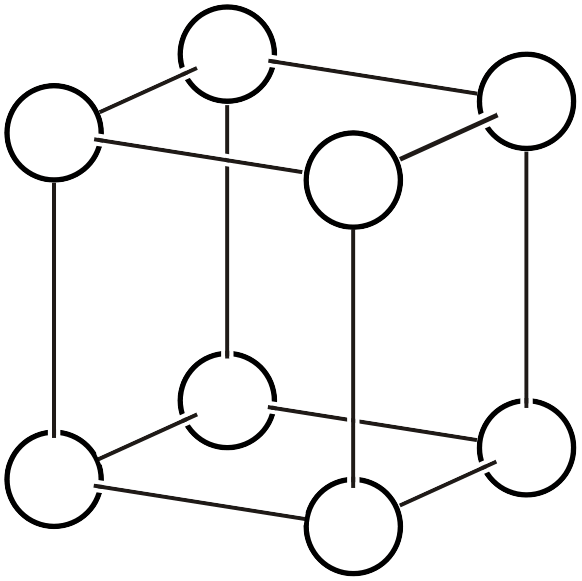 | \(Pm\bar{3}m\) | \(m\bar{3}m\) |
| \(cI\) | 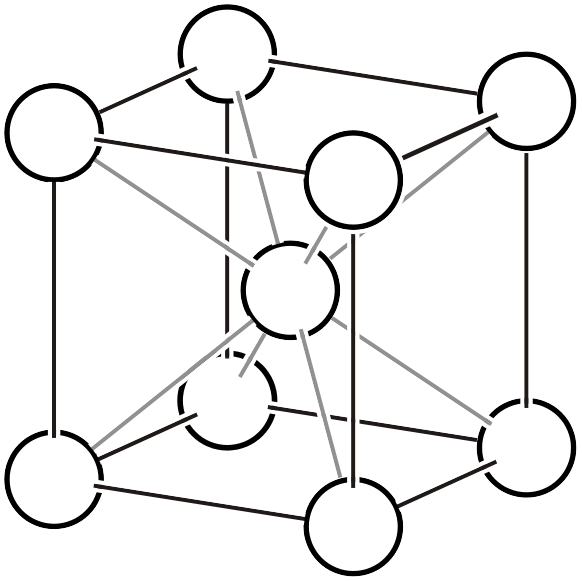 | \(Im\bar{3}m\) | |
| \(cF\) | 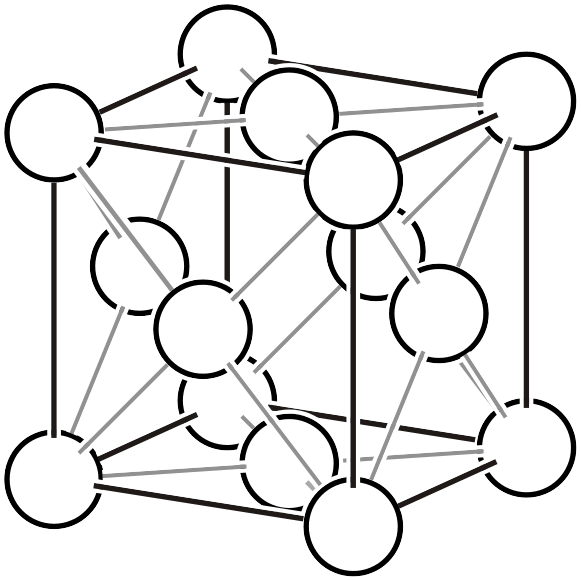 | \(Fm\bar{3}m\) |
You may wonder: if we assumed a primitive lattice, why do centered lattices arise? As explained on the “1.3. Lattice Parameters, Crystal Systems, and Bravais Lattices” page, centered lattices can be decomposed into primitive lattices, so conversely, by assuming a primitive lattice of just the right shape, one can generate a centered lattice.
In real crystals, atoms exist at positions other than the unit cell vertices, but if we focus only on the translational symmetry elements of the space group, every crystal can always be classified into one of the above 14 types. This classification is called the Bravais arithmetic class or simply the Bravais class. Bravais lattice (types) and Bravais arithmetic classes have a one-to-one correspondence. However, to avoid confusion between the two, the convention for Bravais arithmetic classes is to place the lattice symbol from the space group notation at the end, and further convert the base-centered lattice symbol to \(S\), writing for example \(mmmI\) (corresponding to space group \(Immm\)) or \(2/mS\) (corresponding to space group \(C2/m\)).
Lattice System, Crystal System, and Crystal Family
These are terms that are quite easy to confuse. Although not everyone may be troubled by this, let us explain them just in case.
Lattice System
All crystals can be classified into 14 Bravais arithmetic classes (≈ Bravais lattices). The lattice system is a further classification of the Bravais arithmetic classes based on their corresponding point groups. Focusing on a single lattice point within a Bravais lattice, the point group formed by the symmetry operations that keep it fixed4 can be summarized into the following 7 types. The lattice system is the classification of Bravais arithmetic classes according to this criterion.
| Lattice system | Point group | Corresponding Bravais lattices |
|---|---|---|
| Triclinic | \(\bar{1}\) | \(aP\) |
| Monoclinic | \(2/m\) | \(mP\), \(mC\) |
| Orthorhombic | \(mmm\) | \(oP\), \(oC\), \(oI\), \(oF\) |
| Tetragonal | \(4/mmm\) | \(tP\), \(tI\) |
| Rhombohedral | \(\bar{3}m\) | \(hR\) |
| Hexagonal | \(6/mmm\) | \(hP\) |
| Cubic | \(m\bar{3}m\) | \(cP\), \(cI\), \(cF\) |
For all lattice systems except the hexagonal, the symmetry of the unit cell shape directly corresponds to the point group5. However, it would be a mistake to think that lattice systems are classified by the symmetry of the unit cell shape. Under this reasoning, one cannot explain the symmetry of the hexagonal lattice system (point group \(6/mmm\)) unit cell6. Recall that a “lattice” is not a single unit cell (a parallelepiped box) but rather a lattice pattern expressing the relative positional relationships of lattice points. Please note that the lattice system is ultimately a classification based on the symmetry (point group) of the lattice pattern.
Crystal System
All crystals can be classified into 32 (geometric) crystal classes (≈ crystallographic point groups). The crystal system is a further classification of the crystal classes as follows.
| Crystal system | Crystal classes (point groups) | Unique symmetry elements |
|---|---|---|
| Triclinic | \(1\), \(\bar{1}\) | \(1\) or \(\bar{1}\) |
| Monoclinic | \(2\), \(m\), \(2/m\) | \(2\) or \(m\) |
| Orthorhombic | \(222\), \(mm2\), \(mmm\) | \(2\) or \(m\) in 3 directions |
| Tetragonal | \(4\), \(\bar{4}\), \(4/m\), \(422\), \(\bar{4}2m\), \(4/mmm\) | \(4\) or \(\bar{4}\) |
| Trigonal | \(3\), \(\bar{3}\), \(32\), \(3m\), \(\bar{3}m\) | \(3\) or \(\bar{3}\) |
| Hexagonal | \(6\), \(\bar{6}\), \(6/m\), \(622\), \(6mm\), \(\bar{6}m2\), \(6/mmm\) | \(6\) or \(\bar{6}\) |
| Cubic | \(23\), \(m\bar{3}\), \(432\), \(\bar{4}3m\), \(m\bar{3}m\) | \(3\) in four directions |
The crystal system is a more widely used classification than the lattice system, but there is actually no deep mathematical background behind this classification. The first guiding principle for the crystal system classification is to identify commonalities among the various symmetry elements possessed by the 32 crystal classes (and their corresponding point groups) and classify accordingly. For instance, the three point groups \(222\), \(mm2\), \(mmm\), which have symmetry elements \(2\) or \(m\) in 3 directions, are grouped together. You might wonder: why restrict to “3 directions”? Why not allow just 1 direction and include \(2\), \(m\), \(2/m\) in the same group? This is a perfectly reasonable question. So let us add another guiding principle: try to make the classification as close as possible to the lattice system classification7. That is, we consider commonalities in symmetry elements such that space groups belonging to the same lattice system belong, as much as possible, to the same crystal system. Taking this second principle into account leads to the 7 crystal systems shown in the table above.
This classification appears clean at first glance, but there is an important caveat. The trigonal crystal system does not satisfy the second principle. Space groups belonging to the trigonal crystal system can belong to either the hexagonal lattice system or the rhombohedral lattice system, yet their crystal class is the same (for example, space groups \(P3\) and \(R3\) belong to the hexagonal and rhombohedral lattice systems respectively, but both have crystal class \(3\) and are indistinguishable). In this sense, it may be clearer to think of the trigonal crystal system as the collection of crystal classes that can belong to either the hexagonal or rhombohedral lattice system, and the hexagonal crystal system as the collection of crystal classes that always belong to the hexagonal lattice system8. These issues are also discussed in detail on the “Topics in Trigonal/Hexagonal Crystal Systems” page. Aside from the trigonal and hexagonal crystal systems, the remaining 5 crystal systems (triclinic, monoclinic, orthorhombic, tetragonal, and cubic) have a complete correspondence with the lattice systems of the same name (for example, space groups belonging to the cubic crystal system always belong to the cubic lattice system).
Crystal Family
Once you understand the concepts of lattice system and crystal system, understanding the concept of crystal family is straightforward. Essentially, the crystal family is the greatest common divisor of the lattice system and crystal system classifications, where the parts that disagreed between them are combined into the “hexagonal crystal family.” Below, we summarize the relationships among crystal family, lattice system, and crystal system.
| Crystal family | Lattice system | Crystal system | No. of Bravais lattices | No. of point groups | No. of space groups |
|---|---|---|---|---|---|
| Triclinic | 1 | 2 | 2 | ||
| Monoclinic | 2 | 3 | 13 | ||
| Orthorhombic | 4 | 3 | 59 | ||
| Tetragonal | 2 | 7 | 68 | ||
| Hexagonal | Rhombohedral | Trigonal | 1 | 5 | 7 |
| Hexagonal | 1 | 18 | |||
| Hexagonal | 7 | 27 | |||
| Cubic | 3 | 5 | 36 | ||
The hexagonal crystal family requires particular attention. This family contains three types of space groups: “trigonal crystal system in the rhombohedral lattice system,” “trigonal crystal system in the hexagonal lattice system,” and “hexagonal crystal system in the hexagonal lattice system.” It is indeed confusing. Note that classifications such as “trigonal crystal family,” “rhombohedral crystal system,” or “trigonal lattice (system)” do not exist, so be careful not to accidentally use such terms. The other crystal families have a complete correspondence with the lattice system and crystal system of the same name, so there is no need to worry.
Summary
Finally, let us summarize the hierarchical structure of space group classifications introduced on this page. This diagram is a modified version of Fig. 1.3.4.1 from ITA (6th ed).
At the top level sit the 230 space groups — more precisely, space group types. By merging isomorphic duplicates, they are classified into 219 affine space groups. By converting the screw operations and glide operations of affine space groups to rotation operations and mirror operations respectively, 73 arithmetic crystal classes (≈ symmorphic space groups) are obtained.
Focusing on translational operations, arithmetic crystal classes are classified into 14 Bravais arithmetic classes (≈ Bravais lattices), which are further classified into 7 lattice systems based on the symmetry of lattice points. Alternatively, focusing on point group elements, arithmetic crystal classes are classified into 32 geometric crystal classes (≈ crystal family point groups), which are further classified into 7 crystal systems by extracting common symmetry elements.
The classification obtained by taking the greatest common divisor of the lattice system and crystal system is the 6 crystal families.
Footnotes
- In fact, most Japanese textbooks do not distinguish between “space group” and “space group type.” Strictly speaking, if the temperature of a crystal changes by even 1°C, the volume (i.e., the lattice parameters) changes due to thermal expansion, so even though the space group type remains the same, the space group changes. But such strict terminology would be far too cumbersome for practical use. ↩︎
- Crystal class is sometimes translated as “晶族” in Japanese. However, this creates the problem of how to translate crystal family. On this website, we consistently use “結晶類” for crystal class. https://dictionary.iucr.org/Crystal_class ↩︎
- According to IUCr recommendations, since a Bravais lattice represents a type of lattice rather than an individual lattice itself, it is recommended to use the expression “Bravais type of lattice.” Perhaps it would be better to say “Bravais lattice type” in English as well, but for the same reason as with space group “types” above, on this website we simply write “Bravais lattice.” https://dictionary.iucr.org/Bravais_lattice ↩︎
- This can be thought of as the quotient group of the space group, or equivalently as the maximal subgroup. ↩︎
- For the unit cell shape of the rhombohedral lattice, consider not a right prism with a rhombic base (all edges equal, interior angles 60° and 120°) as shown on the left of the figure below, but rather a rhombohedron with three equal edges meeting at equal angles (\(\alpha\)) as shown on the right.
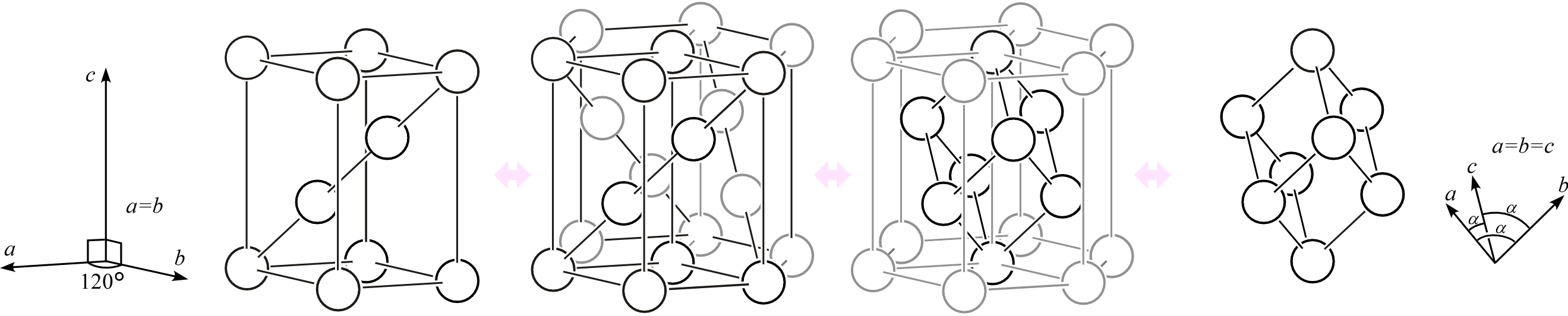 ↩︎
↩︎ - If we explicitly describe the symmetry of the hexagonal lattice unit cell — a right prism with a rhombic base (all edges equal, interior angles 60° and 120°) — its point group is \(mmm\). ↩︎
- Point groups do not contain translational elements, so attempting to create a classification similar to the lattice system, which is premised on translational elements, is inherently somewhat forced. This is the source of the irregularity in the hexagonal crystal family. While crystal systems have some significance when describing the shape of finite objects such as molecules, the concept of lattice systems is more important when dealing with crystals, which necessarily contain translational elements. In my personal opinion, researchers studying crystals may benefit more from focusing on lattice systems rather than crystal systems. ↩︎
- You might think the difference between the trigonal and hexagonal crystal systems is simply whether the order of rotation or rotoinversion is “3” or “6,” but it is not that straightforward. The hexagonal point group \(\bar{6}\) is equivalent to the expression \(3/m\), so one cannot judge by order alone. Furthermore, if one considers rotoreflection symmetry elements instead of rotoinversion (\(\bar{3} = S_6\), \(\bar{6} = S_3\)), the apparent order changes. ↩︎