Lattice Parameters and Vectors
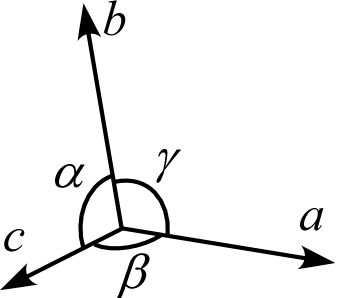
The lattice of a three-dimensional crystal is usually expressed using three lattice vectors \(\textbf{a}, \textbf{b}, \textbf{c}\) with respective lengths \(a,b,c\) and the angles between them \(\alpha,\beta,\gamma\). These six parameters—\(a,b,c,\alpha,\beta,\gamma\)—are called the lattice parameters (cell constants, lattice constants). Note that except for cases with special symmetry, the lengths \(a,b,c\) are not necessarily equal, and the angles \(\alpha,\beta,\gamma\) are not necessarily \(90^\circ\).
Representation of Direction in Crystals
Lattice vectors are not necessarily orthogonal and their lengths are not necessarily equal. In crystallography, a direction (lattice direction) corresponding to such a coordinate system is denoted using square brackets \([\,]\) as \([u\,v\,w]\), called direction indices. \([u\,v\,w]\) is defined as the vector \(u\mathbf{a}+v\mathbf{b}+w\mathbf{c}\). In other words, \([u\,v\,w]\) represents coordinates in a vector space with \(\mathbf{a}, \mathbf{b}, \mathbf{c}\) as the basis, and \(\mathbf{a}, \mathbf{b}, \mathbf{c}\) are called basis vectors.
\(u,v,w\) can be any real numbers, but when they consist only of integers, the vector connects lattice point to lattice point and thus always has the property of a “translation” vector (for example, \([100]\) is \(\mathbf{a}\) itself, and \([211]\) is \(2\mathbf{a}+\mathbf{b}+\mathbf{c}\)). When representing negative numbers, it is preferable to place a bar over the digit rather than use a minus sign1 (for example, \([1\bar{1}1]\)).
Depending on the symmetry of the crystal, multiple directions can be equivalent. For example, if \([100]\) and \([010]\) are crystallographically equivalent, they are expressed using angle brackets \(\langle\,\rangle\) as \(\langle100\rangle\). \(\langle100\rangle\) does not represent a single vector but rather a family of equivalent directions (a family of equivalent directions) that includes \([100]\). Naturally, angle brackets are used only in contexts where the material’s symmetry (crystal system or point group) is limited.
Another concept for expressing direction and distance in crystals is the “lattice plane.” This will be explained in detail in “4.1. Lattice Planes and Diffraction“.
Crystal System
The crystal system is one method of classifying crystals, based on the shapes compatible with the symmetry (other than translation) that the crystal possesses. To accurately understand this classification requires knowledge of space groups, so that discussion will be deferred; for now, proceed with the understanding that three-dimensional crystal systems are classified as follows.
| Crystal System | Triclinic | Monoclinic | Orthorhombic | Trigonal Hexagonal | Tetragonal | Cubic |
| Description | Three edges do not intersect orthogonally | One edge (\(b\)) of the three edges is orthogonal to the other two | All three edges are mutually orthogonal | Two edges (\(a,b\)) intersect at 120° with equal lengths, and are orthogonal to the remaining edge (\(c\)) | Three edges are orthogonal, and two of them (\(a,b\)) have equal lengths | Three edges are orthogonal with all equal lengths |
| Schematic Diagram |  | 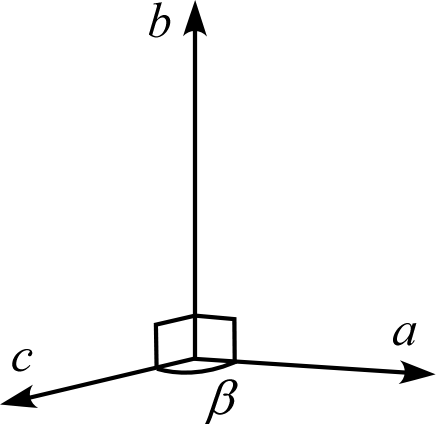 | 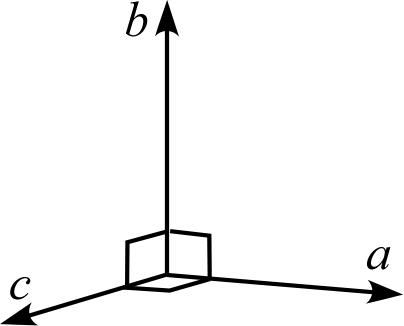 | 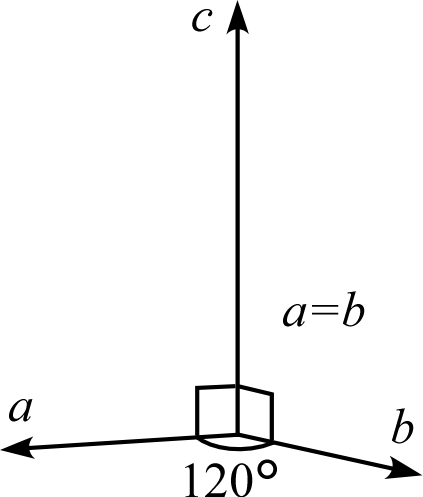 | 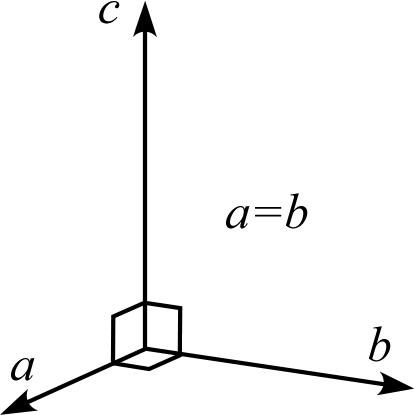 | 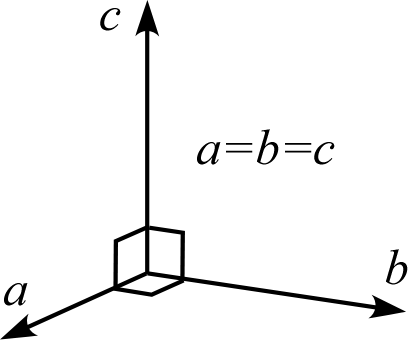 |
| Unique Symmetry Elements2 | \(1\) or \(\bar{1}\) | \(2\) or \(m\ (=\bar{2})\) | Three directions with \(2\) or \(m\ (=\bar{2})\) | \(3\) or \(6\) | \(4\) or \(\bar{4}\) | Four directions with \(3\) |
Each crystal system has unique symmetry elements (rotation axes or rotoreflection axes) that characterize it, and the direction of these elements is called the principal axis. When there are two or more unique symmetry elements, the second and subsequent ones are called secondary axes. This topic will be discussed again in “2.3. Notation of Point Groups and Space Groups“.
Here, we introduce some trivia topics regarding crystal systems.
Why Is There No Diclinic Crystal System?
Why are there triclinic and monoclinic crystal systems, but no “diclinic” system? If “diclinic” were interpreted literally, it would be a crystal lattice where \(\alpha\) is \(90°\) but \(\beta\) and \(\gamma\) are not. Such a parallelepiped might seem to have higher symmetry compared to the triclinic case. However, upon closer examination, the only operations that leave this shape invariant are a 1-fold rotation or 1-fold rotoreflection; no rotation or rotoreflection operations of order 2 or higher are allowed. In other words, from the perspective of symmetry, it is the same as the triclinic system. Even if \(\alpha\) happens to be \(90°\), the crystal remains triclinic.
Difference Between Trigonal and Hexagonal Crystal Systems
Have you ever wondered why these two must be distinguished despite having the same unit cell shape? Details have been written on the “6.1. Topics on Trigonal/Hexagonal Crystal Systems” page, so please refer to it.
Tetragonal and Cubic Systems and Four-fold Rotation
Try imagining the shape of crystals belonging to tetragonal or cubic systems. Because the words “tetragonal” or “cubic” are included, one cannot help but imagine shapes like squares or cubes—that is, shapes that include four-fold rotation operations. Of course, there are such crystals, but there are also cases without any square-like elements. For example, in the case of the point group \(23\) belonging to the cubic system, the following crystal shape is allowed. This crystal is surrounded by six (100) faces (red) and four (111) faces (green).
Note that tetragonal or cubic systems do not necessarily contain four-fold rotation operations.
Primitive Lattice and Complex Lattice
First, note that white circles ◯ in the figures in this section represent lattice points; no concrete material like atoms actually exists. What is important is the direction and distance of the translation vectors connecting lattice point to lattice point.
A unit cell is a parallelepiped with three independent translation vectors in space as its three edges. A primitive lattice is a crystal lattice in which the translation vectors are chosen so that the volume of the parallelepiped is minimized. In a primitive lattice, each vertex of the parallelepiped is a lattice point, and there are no other lattice points.
On the other hand, a complex lattice is a crystal lattice in which a larger volume is deliberately chosen instead of the minimum. In a complex lattice, lattice points exist at each vertex as well as at face centers or the center of the lattice. Why is a larger volume deliberately chosen? This is because priority is given to classifying crystal lattice shapes by “crystal system” and the mathematical expression is to be simplified3.
Primitive Lattice (\(P\))
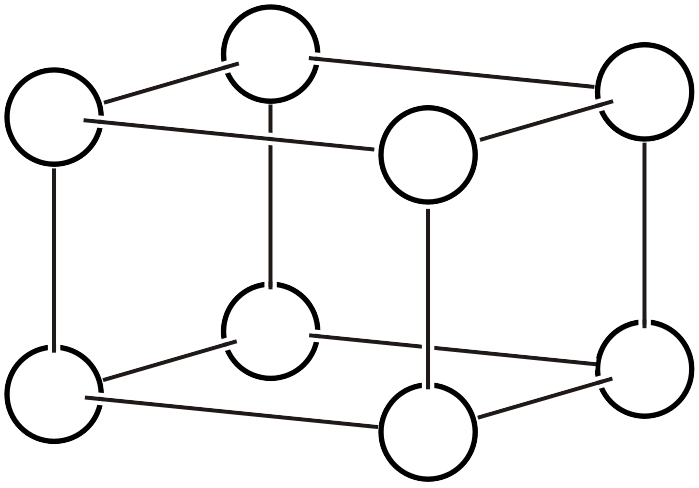
Complex lattices are lattices deliberately chosen with larger volume in consideration of crystal systems, but if an arbitrarily large volume is chosen, the utility is actually lost. Meaningful ways to define complex lattices are limited to the following four types.
- Body-centered lattice (\(I\)) : A crystal lattice with a lattice point at the center of the unit cell
- Base-centered lattice (\(A, B, C\)) : A crystal lattice with a lattice point at the center of a face of the unit cell
- Face-centered lattice(\(F\)): A crystal lattice with lattice points at the centers of all faces of the unit cell
- Rhombohedral lattice(\(R\)): A crystal lattice with lattice points at positions that trisect the body diagonal of the unit cell (trigonal system only)
Body-centered lattice
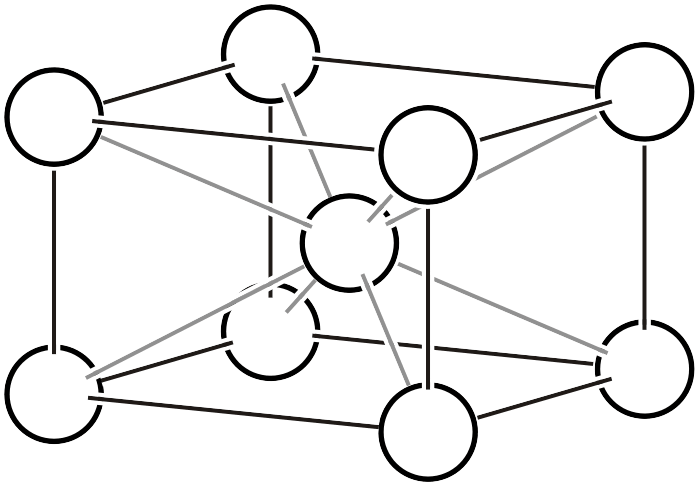
Base-centered lattice
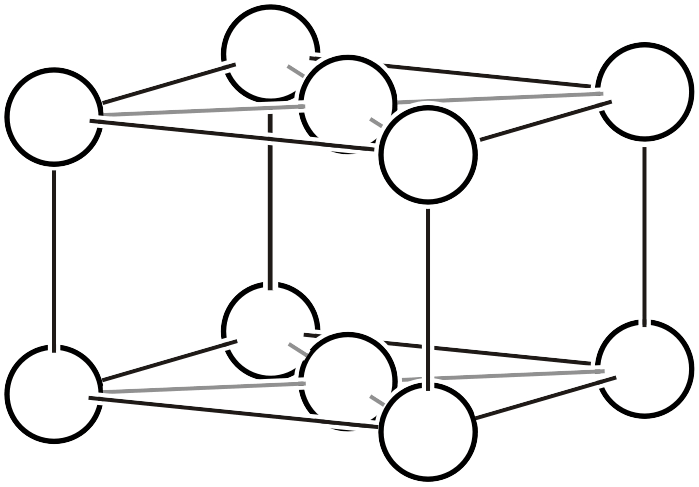
Face-centered lattice
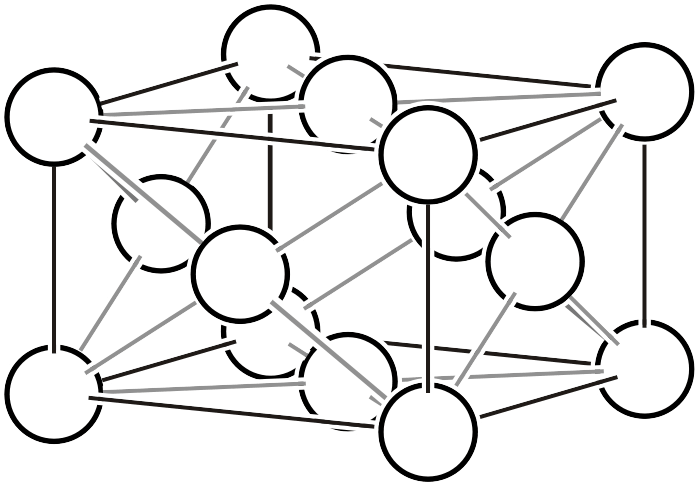
Rhombohedral lattice

Next, let us examine the geometric relationships between each complex lattice and the primitive lattice.
Body-centered Lattice (\(I\)4)
The body-centered lattice contains two lattice points (white circles) within the lattice. In the left figure, a total of 9 lattice points are drawn, but remember that the lattice points at the vertices contribute only 1/8 to this lattice. Also note that the positions of lattice points and atoms do not necessarily coincide. The body-centered lattice is deliberately chosen with a larger volume and can be transformed into a primitive lattice. As shown in the right figure, if four body-centered lattices are arranged and a lattice indicated by thick black lines is chosen, it becomes a primitive lattice with half the volume.
Body-centered lattice

Body-centered lattice ➡ Primitive lattice

Base-centered Lattice (\(A, B, C\))
The base-centered lattice contains two lattice points (white circles) within the lattice. As shown in the left figure, among the six faces of the unit cell, lattice points exist at the center of each of the two opposite faces. As shown in the right figure, if two base-centered lattices are arranged and a lattice indicated by thick black lines is chosen, the unit cell has half the volume.
Base-centered lattice

Base-centered lattice ➡ Primitive lattice
The symbol for base-centered lattice has three types: \(A, B, C\), representing the relationship with axes \(a,b,c\). For example, base-centered lattice \(A\) represents a lattice with a lattice point at \(b/2+c/2\).
Face-centered Lattice (\(F\))
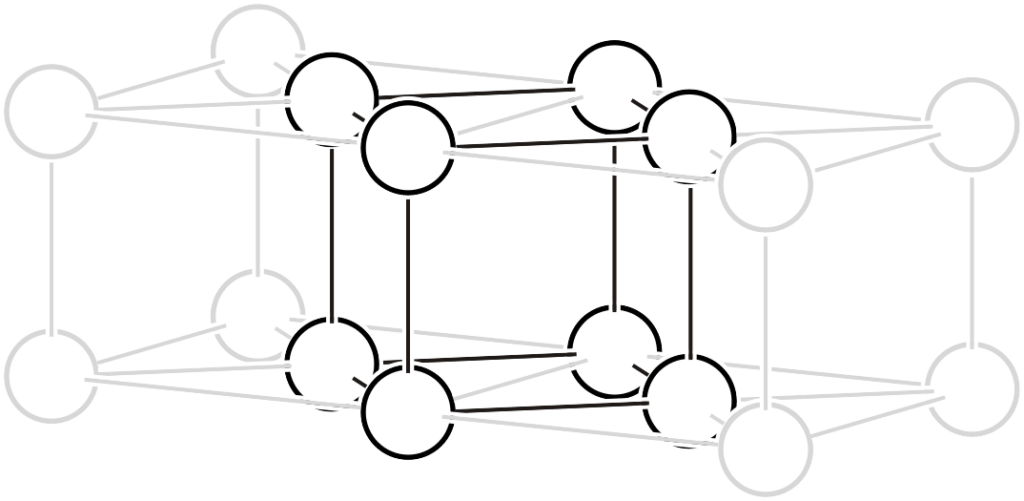
The face-centered lattice contains four lattice points (white circles) within the lattice. As shown in the left figure, lattice points exist at the centers of all faces of the unit cell. As shown in the right figure, if a lattice indicated by thick black lines is chosen, it becomes a unit cell with 1/4 of the volume.
Face-centered lattice

Face-centered lattice ➡ Primitive lattice
Rhombohedral Lattice (\(R\))
The rhombohedral lattice is a term used for crystals belonging to some trigonal systems, but care must be taken as its meaning varies depending on context. When simply saying “rhombohedral lattice,” it often means “a crystal lattice with equal edge lengths and equal angles (\(a=b=c,\,\, \alpha=\beta=\gamma\))”, but here we use it to mean “a crystal lattice expressed in hexagonal setting (\(a=b, \alpha=\beta=90°, \gamma=120°\))” for crystals classified in the “rhombohedral system5“. It can also be called a rhombohedral hexagonal lattice.
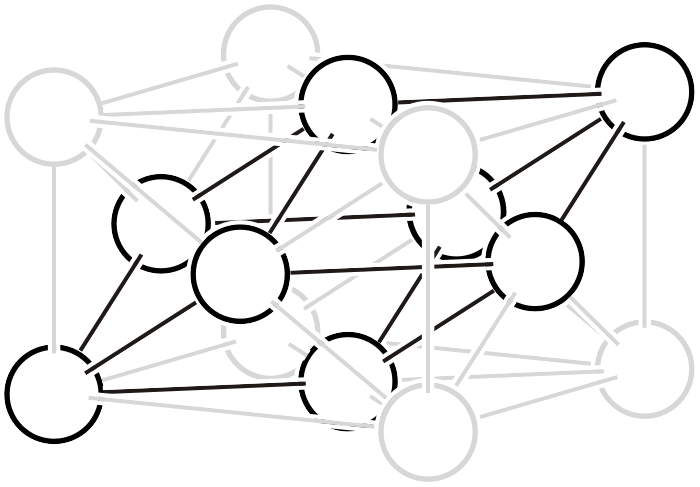
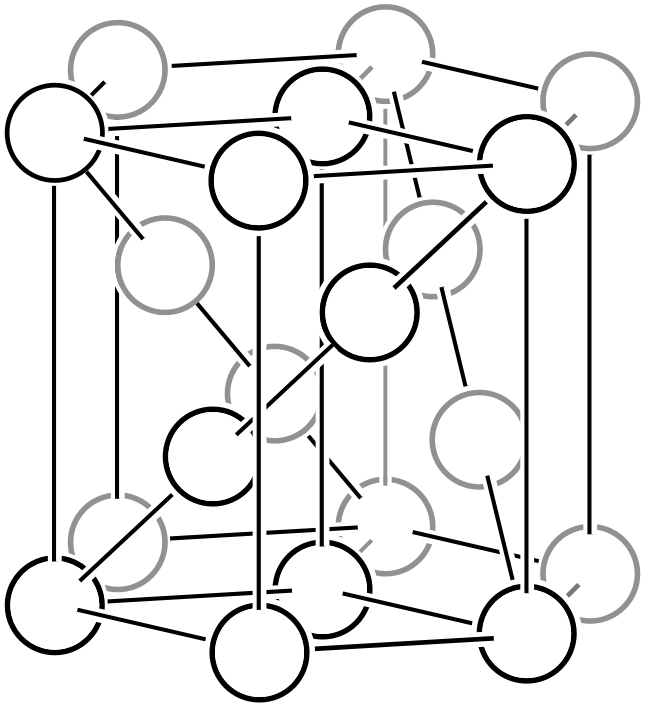
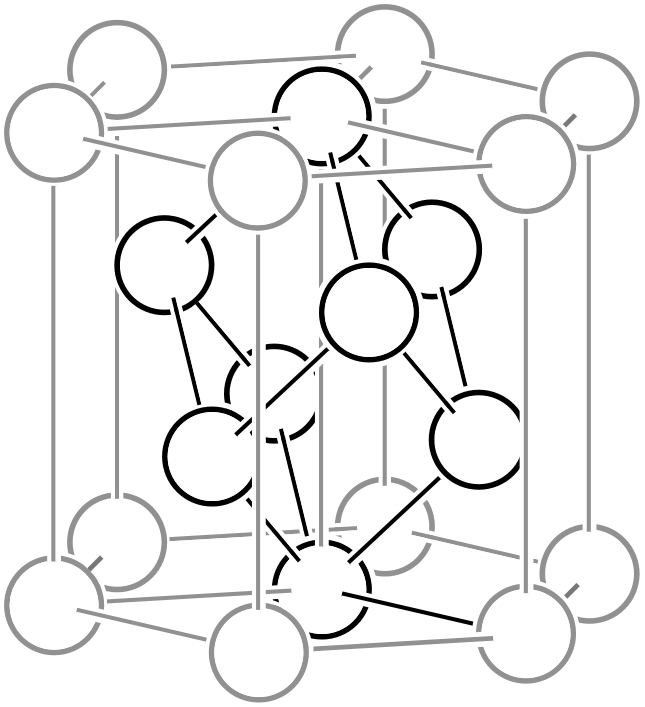
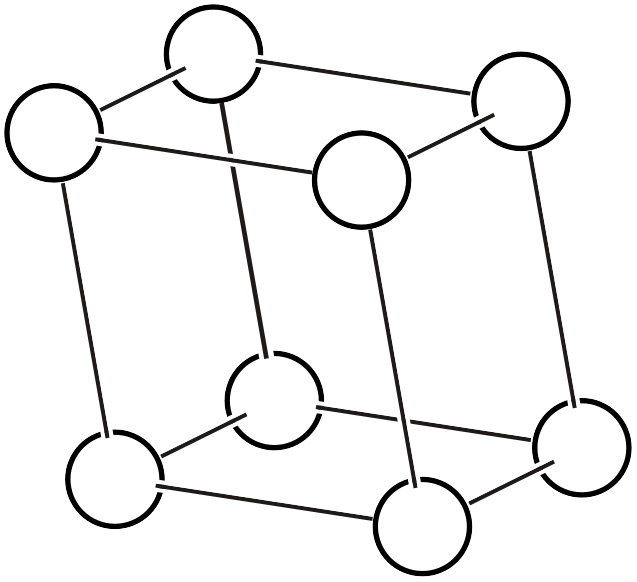
As shown in the left figure, the rhombohedral hexagonal lattice has lattice points at positions that trisect the direction of the body diagonal of the unit cell, containing three lattice points (white circles) within the lattice. Like other complex lattices, the rhombohedral hexagonal lattice can be transformed into a primitive lattice by arranging several lattices. First, since the trigonal system always has a 3-fold rotation axis, let us arrange three rhombohedral hexagonal lattices rotated by 120° as shown in the middle figure. By connecting all the lattice points located at the centers of the three lattices and those at heights of 1/3 and 2/3, a primitive lattice with 1/3 of the volume (that is, the rhombohedral setting of the rhombohedral system, or simple rhombohedral lattice) can be obtained. Please also refer to the description on the “6.1. Topics on Trigonal/Hexagonal Crystal Systems” page for details on rhombohedral lattice geometry.
Rhombohedral hexagonal lattice

Three rhombohedral hexagonal lattices in 120° relation
Primitive lattice (simple rhombohedral lattice)
Bravais Lattice6
How many types of crystal lattices with atoms only at lattice points can be classified from the perspective of symmetry (how many non-isomorphic groups are there)? This is the concept of the Bravais lattice7. In other words, it is the space lattice obtained by excluding those that do not lose symmetry even when using a smaller unit cell, or those that become equivalent through transformation of basis vectors, from the space lattice combining all complex lattice elements for all crystal systems. Detailed discussion of this will be done on the “5.1. Classification of Space Groups” page; for now, let us proceed. There are 5 Bravais lattices in two dimensions and 14 in three dimensions. Historically, the concept of Bravais lattice was derived first, and the classification of crystal systems was done later, but here we explain each Bravais lattice (three-dimensional) for each crystal system.
Triclinic System
The triclinic system has only a primitive lattice as its Bravais lattice. This is because the symmetry operations (linear operations) excluding translation in the triclinic system are only \(1\) or \(\bar{1}\), and deliberately taking body-centered or face-centered lattices yields no benefit. However, in limited situations (for example, when one wants to maintain the unit cell shape before and after a phase transition), it is sometimes done deliberately.

Primitive triclinic lattice (\(aP\))
Monoclinic System
The monoclinic system normally has a primitive lattice and a base-centered lattice with the \(b\) axis as the principal axis. Body-centered and face-centered monoclinic lattices become equivalent to the base-centered lattice through appropriate axis transformation, so they are not classified separately. However, in some cases, a different axis is used as the principal axis, or a body-centered lattice is used instead of a base-centered lattice. Such transformations are described on the “6.3. Choice of Axes, Axis Transformation” page.

Primitive monoclinic lattice (\(mP\))
Base-centered monoclinic lattice (\(mC\))
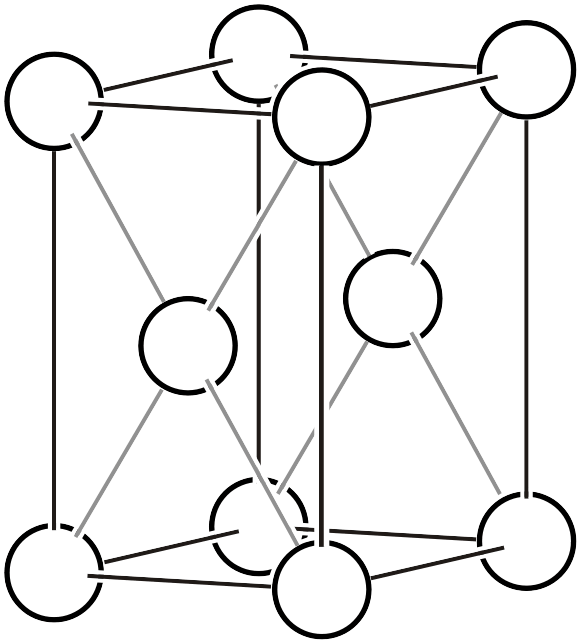
Orthorhombic System
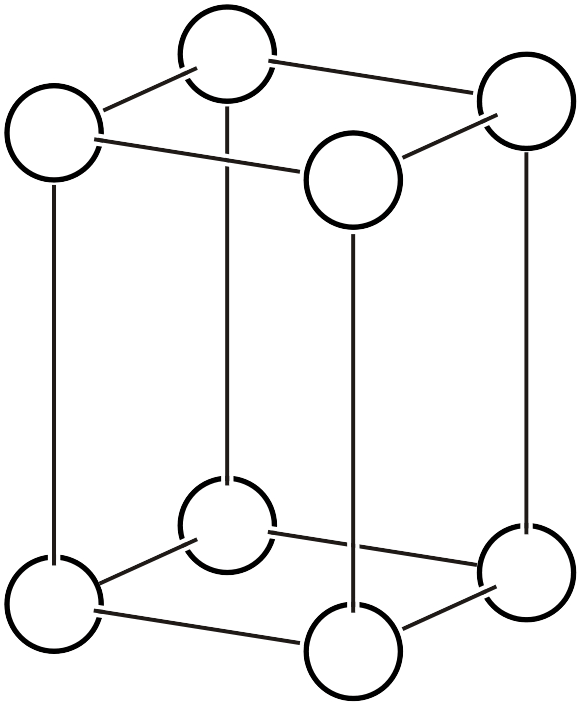
The orthorhombic system has all four types of Bravais lattices: primitive, base-centered, body-centered, and face-centered.

Primitive orthorhombic lattice (\(oP\))

Body-centered orthorhombic lattice (\(oI\))

Base-centered orthorhombic lattice (\(oC\))

Face-centered orthorhombic lattice (\(oF\))

Trigonal System, Hexagonal System
The trigonal and hexagonal systems have a primitive lattice and a rhombohedral lattice with the \(c\) axis as the principal axis. For details on rhombohedral lattice geometry, please also refer to the “6.1. Topics on Trigonal/Hexagonal Crystal Systems” page.

Primitive hexagonal lattice (\(hP\))
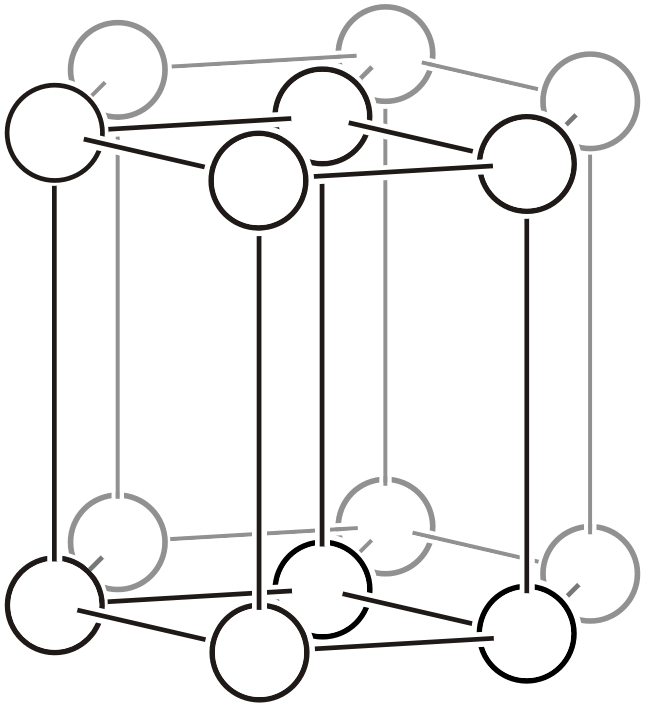
Rhombohedral hexagonal lattice (\(hR\))
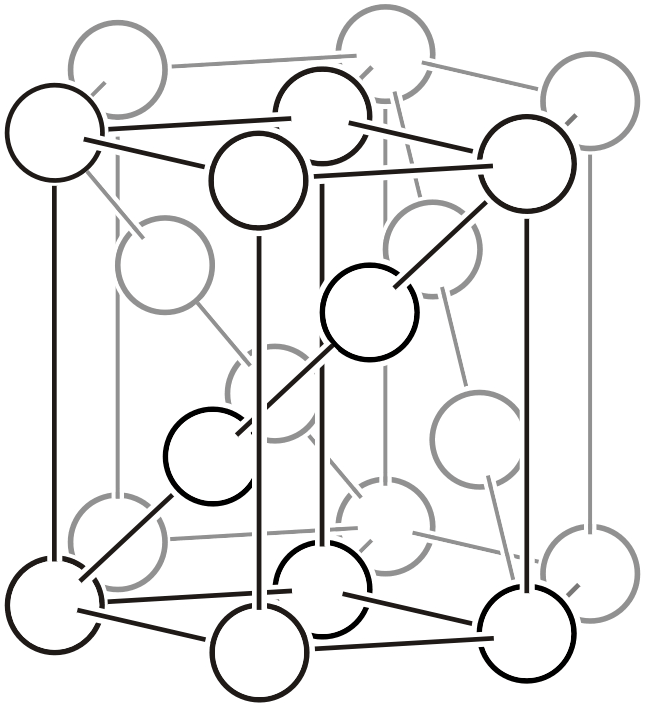
Tetragonal System
The tetragonal system has a primitive lattice and a body-centered lattice with the \(c\) axis as the principal axis. The base-centered tetragonal lattice can be re-expressed as a primitive tetragonal lattice and is not classified. The face-centered tetragonal lattice can be re-expressed as a body-centered tetragonal lattice and is not classified.

Primitive tetragonal lattice (\(tP\))
Body-centered tetragonal lattice (\(tI\))
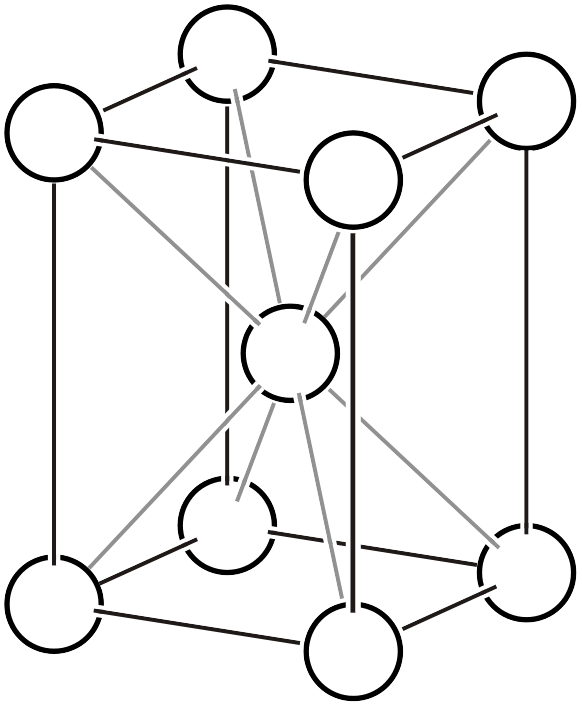
Cubic System
The cubic system has a primitive lattice, body-centered lattice, and face-centered lattice. The base-centered cubic lattice does not exist because it contradicts the 3-fold rotation operation existing in the [111] direction.

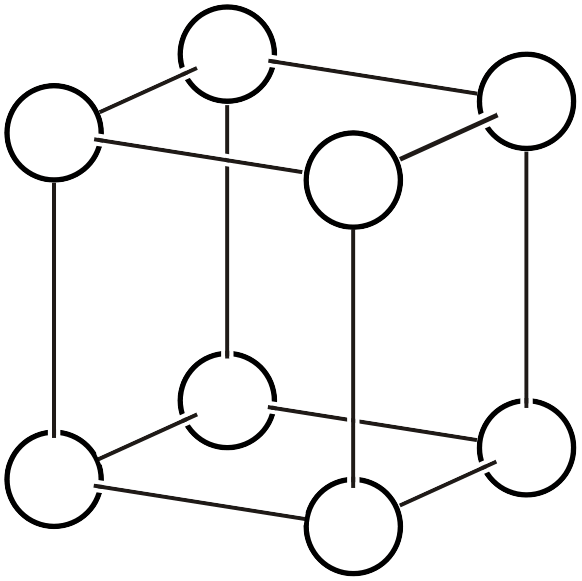
Primitive cubic lattice \(cP\)
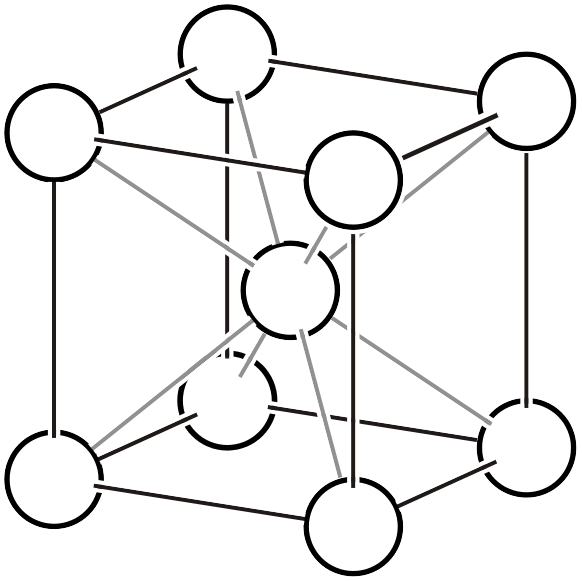
Body-centered cubic lattice \(cI\)
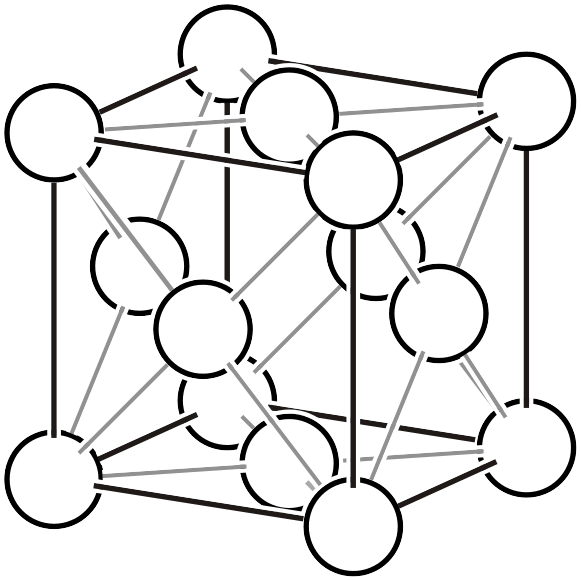
Face-centered cubic lattice \(cF\)
Footnotes
- At first glance it may look like a rotoinversion symbol, but it is entirely different, so be careful. ↩︎
- Here we have listed only rotation and rotoinversion, but in fact glide and screw can also be unique elements. A glide is an order-2 rotoinversion (=\(m\)) with translation added, so it is treated equivalently to \(m\). A screw directly corresponds to the rotation order, so for example, if \(2_1\) screw axes exist in three directions, the crystal system is orthorhombic. ↩︎
- If the concept of complex lattices were not introduced, situations would arise such as “a crystal with a 2-fold rotation axis that cannot take a right-angled prismatic unit cell,” making it very laborious (though not impossible) to understand the crystal’s structure and physical properties. ↩︎
- Derived from the German word innenzentriert (internally centered). In English it would be body centered, so one might prefer the letter “B,” but that would conflict with the symbol for the base-centered lattice. ↩︎
- For lattice systems, see “5.1. Classification of Space Groups“. ↩︎
- Named after Auguste Bravais, a 19th-century French crystallographer. ↩︎
- In this sense, the white circles in the figures can be considered atoms rather than lattice points, but the importance of the Bravais lattice concept lies not in “crystals where atoms exist only at lattice points” themselves, but in the classification (Bravais arithmetic class) that maps all crystals to Bravais lattices. A detailed explanation will be given in “5.1. Classification of Space Groups“. ↩︎